Associate Professor, University of Maryland Eastern Shore, MD
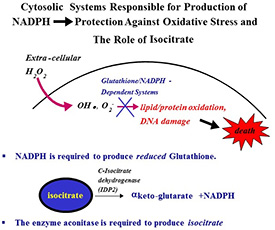
Our straightforward question is to ascertain what metabolic genes are turned on and off during oxidative stress. In addition, it is known that P. marinus is resistant to killing by H2O2, so this study may yield clues as to how this parasite is able to resist oxidative stress. Finally, cytosolic aconitase is regulated by protein kinase C (PKC) in mammalian systems, altering its enzymatic function. A conserved PKC phosphorylation site is present in the P. marinus aconitase, and several PKC orthologues are present in the P. marinus genome. Thus, beyond the various enzymatic pathways that are almost certain to be differentially regulated, we are also very curious to determine what signaling protein orthologues will be stimulated during oxidative stress.
RNA will be generated from cells grown under two different growth regimens. Growth condition #1 will use a standard growth media that is used to cultivate cells, and in which we were able to generate RNA for a study on spliced leader RNA from P. marinus. Growth media #2 will be contain H2O2 at a concentration of 10 mM, which is known to induce oxidative stress in cells. Our straightforward question is to ascertain what metabolic genes are turned on and off during these distinct conditions. In addition, it is known that P. marinus is resistant to killing by H2O2, so this study may yield clues as to how this parasite is able to resist oxidative stress. Finally, cytosolic aconitase is regulated by protein kinase C (PKC) in mammalian systems, altering its enzymatic function. A conserved PKC phosphorylation site is present in the P. marinus aconitase, and several PKC orthologues are present in the P. marinus genome. Thus, beyond the various enzymatic pathways that are almost certain to be differentially regulated, we are also very curious to determine what signaling protein orthologues will be stimulated during oxidative stress.
At UMES I am responsible for teaching students at all levels, including introductory biology for majors, a sophomore level course in genetics, and a junior/senior level course in cell biology. In spring of 2016 I will be teaching a new course in molecular genetics. I will assign a semester project based upon this proposed study. The laboratory portion of the course will include both training in bioinformatics methods and in various cloning procedures. The RNAseq analysis will be integrated into the course through a unit describing bioinformatics approaches.